
So keep this in mind when we're confronting different types of transition metals. They are transition metals, but they're not type two medals because they don't have multiple charges. And because of that, they're characterizes being Type two medals, some of the transition metals ones in red. Ah, lot of them have multiple possible charges. Both of them will be plus two when they do have a charge. They're both in the same group again, so they're gonna be similar to each other. So silver, when it's an ion, is gonna be plus one cadmium and zinc. 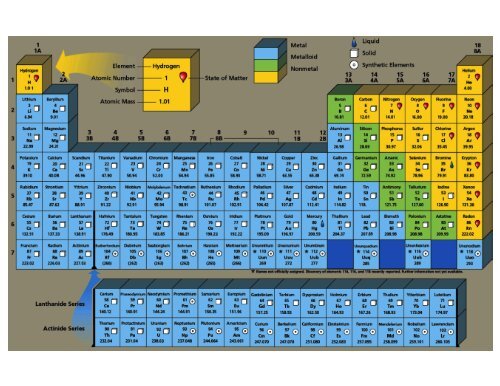
But just realize here that these transition metals or called transition metals because they have a bunch of possible charges now besides the elements in Group three B or three, we also have silver, cadmium and zinc, although their transition metals as well they also have Onley one particular charge. The table is available for download in PDF format for offline printing. This periodic table contains the number, symbol, name, atomic mass and most common valence charge of each element. Now, the way we're able to tell which one of these charges manganese will have will be dependent on the other element is connected to We learn about that later on. The improved table can be found at Black and White Printable Periodic Table Chart with Oxidation States. But then, of course, when you look at other transition metals, you're going to see a bunch of charges like Mangga Knees, for example, could be plus two plus three plus four plus five or even plus seven. And there's some similar chemical properties going on for elements within that group because Skandia Miz plus three, that means the other metals that are in this group with it are also plus three. Note that CuO has only one copper ion, while Cu 2 O has two copper ions. Based on position on the periodic table, the oxide ion has a charge of 2. So, for example, Skandia Mom, which is in Group three or three B, it's plus three. Write the name leaving room for a Roman numeral, since copper forms ions with variable charges. And what we need to realize is that although many of them have multiple charges, there are quite a few that do possess only one charge. So here, if we take a look, we have some of the most common types of transition metals. So just remember, when it comes to type two medals, a majority of them are the transition metals. Now there's gonna be mawr advanced explanations for this later on, but we'll discuss them in much later chapters. Now we're gonna say most transition metals have varying positive charges because of their electron arrangements around the nucleus. So remember Type two medals are metals that possess multiple charges. ION CHARGE TRENDS Alkali Metals always have a +1 Ion Charge Alkaline Earth Metals all have a +2 Ion Charge Nitrogen and Phosphorus both have a -3 Ion Charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
